
Virtual Screening Against Carbohydrate-Binding Proteins: Evaluation and Application to Bacterial Burkholderia ambifaria Lectin | Journal of Chemical Information and Modeling

How To Optimize Shape-Based Virtual Screening: Choosing the Right Query and Including Chemical Information | Journal of Chemical Information and Modeling
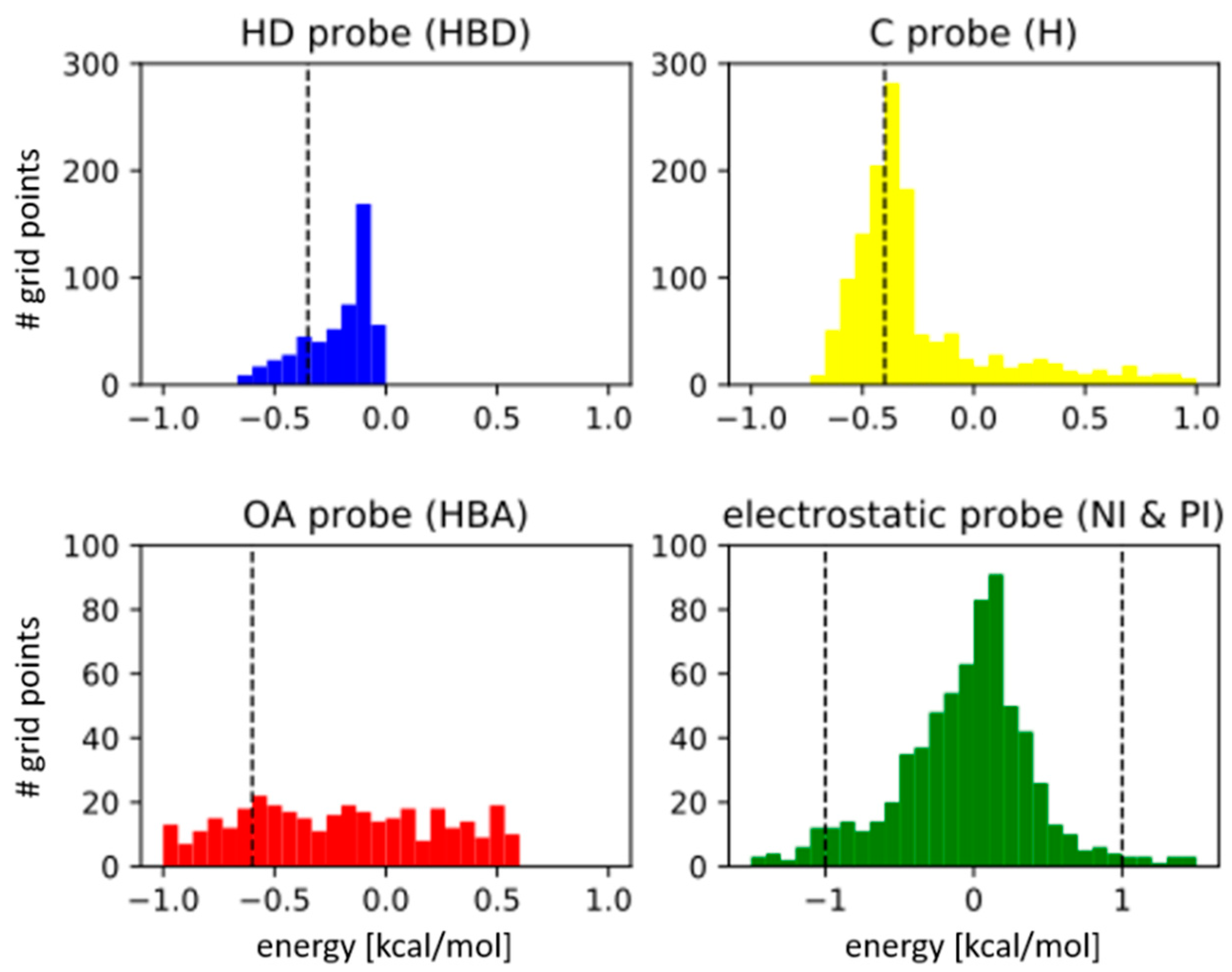
Molecules | Free Full-Text | Truly Target-Focused Pharmacophore Modeling: A Novel Tool for Mapping Intermolecular Surfaces

ANALYSIS OF CARBOHYDRATES AND GLYCOCONJUGATES BY MATRIX‐ASSISTED LASER DESORPTION/IONIZATION MASS SPECTROMETRY: AN UPDATE FOR 2015–2016 - Harvey - 2021 - Mass Spectrometry Reviews - Wiley Online Library

Virtual Screening Against Carbohydrate-Binding Proteins: Evaluation and Application to Bacterial Burkholderia ambifaria Lectin | Journal of Chemical Information and Modeling

PDF) Anthocyanin composition, antioxidant efficiency, and -amylase inhibitor activity of different Hungarian sour cherry varieties (Prunus cerasus L.)
Flavonoids as dual-target inhibitors against α-glucosidase and α-amylase: a systematic review of in vitro studies

Extending the investigation of 4-thiazolidinone derivatives as potential multi-target ligands of enzymes involved in diabetes mellitus and its long-term complications: A study with pancreatic α-amylase - ScienceDirect
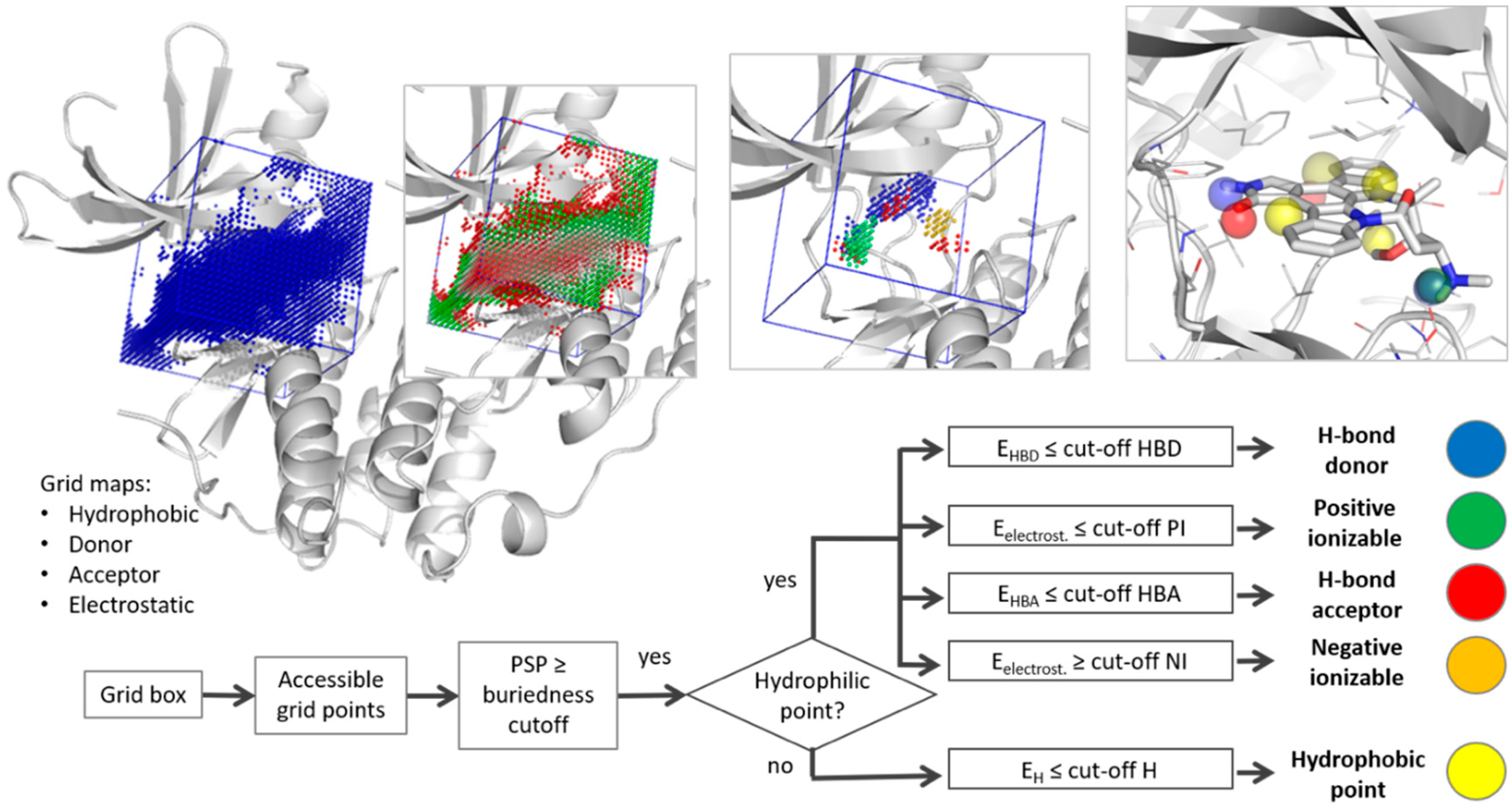
Molecules | Free Full-Text | Truly Target-Focused Pharmacophore Modeling: A Novel Tool for Mapping Intermolecular Surfaces
Flavonoids as dual-target inhibitors against α-glucosidase and α-amylase: a systematic review of in vitro studies

α-Glucosidase inhibitory effect of anthocyanins from Cinnamomum camphora fruit: Inhibition kinetics and mechanistic insights through in vitro and in silico studies - ScienceDirect

Multicomponent reaction for the synthesis of new 1,3,4-thiadiazole-thiazolidine-4-one molecular hybrids as promising antidiabetic agents through α-glucosidase and α-amylase inhibition - ScienceDirect

How To Optimize Shape-Based Virtual Screening: Choosing the Right Query and Including Chemical Information | Journal of Chemical Information and Modeling
Flavonoids as dual-target inhibitors against α-glucosidase and α-amylase: a systematic review of in vitro studies
Flavonoids as dual-target inhibitors against α-glucosidase and α-amylase: a systematic review of in vitro studies

How To Optimize Shape-Based Virtual Screening: Choosing the Right Query and Including Chemical Information | Journal of Chemical Information and Modeling
PDF) Extending the investigation of 4-thiazolidinone derivatives as potential multi-target ligands of enzymes involved in diabetes mellitus and its long-term complications: A study with pancreatic α-amylase

ANALYSIS OF CARBOHYDRATES AND GLYCOCONJUGATES BY MATRIX‐ASSISTED LASER DESORPTION/IONIZATION MASS SPECTROMETRY: AN UPDATE FOR 2015–2016 - Harvey - 2021 - Mass Spectrometry Reviews - Wiley Online Library

ANALYSIS OF CARBOHYDRATES AND GLYCOCONJUGATES BY MATRIX‐ASSISTED LASER DESORPTION/IONIZATION MASS SPECTROMETRY: AN UPDATE FOR 2015–2016 - Harvey - 2021 - Mass Spectrometry Reviews - Wiley Online Library

Multicomponent reaction for the synthesis of new 1,3,4-thiadiazole-thiazolidine-4-one molecular hybrids as promising antidiabetic agents through α-glucosidase and α-amylase inhibition - ScienceDirect

How To Optimize Shape-Based Virtual Screening: Choosing the Right Query and Including Chemical Information | Journal of Chemical Information and Modeling
ANALYSIS OF CARBOHYDRATES AND GLYCOCONJUGATES BY MATRIXâ•'ASSISTED LASER DESORPTION/IONIZATION MASS SPECTROMETRY: AN UPDATE
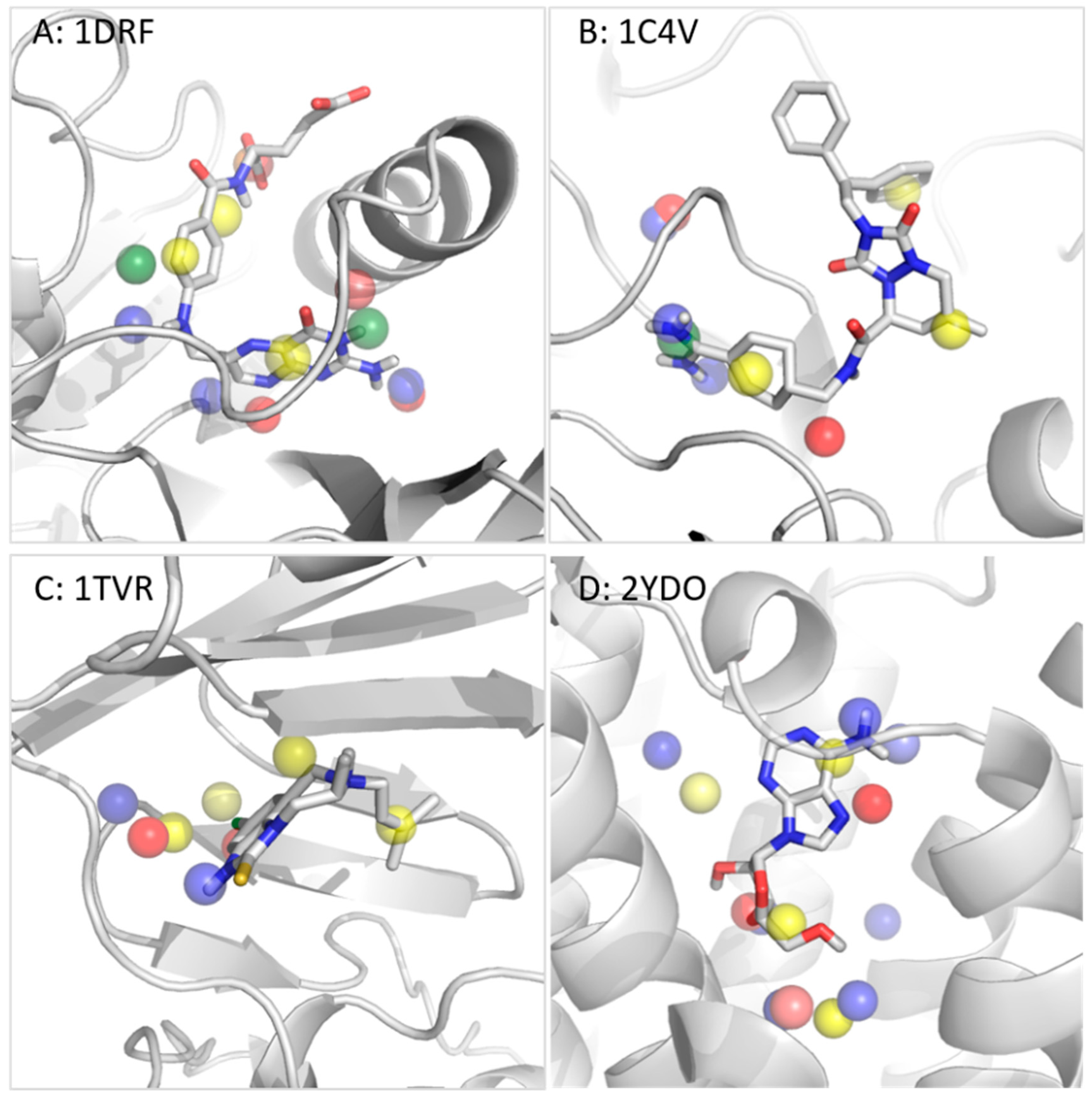
Molecules | Free Full-Text | Truly Target-Focused Pharmacophore Modeling: A Novel Tool for Mapping Intermolecular Surfaces
Flavonoids as dual-target inhibitors against α-glucosidase and α-amylase: a systematic review of in vitro studies

